How Many Neutrons Does a Normal Hydrogen Atom Have
Trace elements make up a _____ percentage of the elements found in living organisms and are _____ for normal metabolic function. Hydrogen has no neutrons just a single proton and a single electron.

Hydrogen Preparation Isotops Hydrogen Uses And Properties
What compounds does Argon form.

. Hydrogen has a mass of 1 because of the mass of its single proton. - small - critical. How many protons electrons and neutrons does water have.
The most common isotope of uranium has 92 protons and 146 neutrons. So H2O consists of a total of 10 protons 8 neutrons and 10 electronsApr 3 2018. So H2O consists of a total of 10 protons 8 neutrons and 10 electrons.
Its atomic number is 14 and its atomic mass is 28. Hydrogen nucleus does not contain any neutron. The electrons have been known to wander.
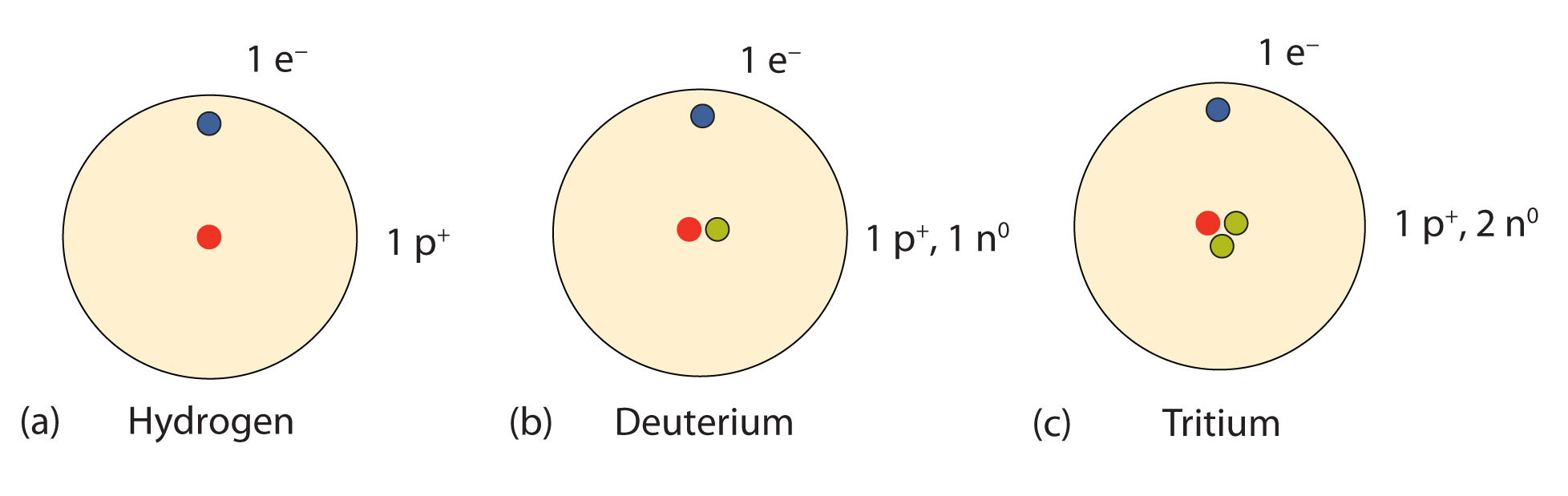
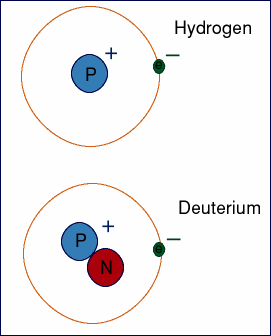
Deutrium or heavy hydrogen contains one neutron. How Many Neutrons Does Water Have. All atomic nuclei contain one or more neutrons except hydrogen-1 which has just a single proton as its nucleus.
Hydrogen - Protons - Neutrons - Electrons - Electron Configuration. Most usually a hydrogen atom has one proton and one electron. Hydrogen has no neutron deuterium has one and tritium has two neutrons.
Sometimes the hydrogen atom has a neutron deuterium. The mass numbers of hydrogen isotopes are 1 2 and 3 respectively. Atoms of these isotopes have one electron to balance the charge of one proton.
It is denoted by the O the O in H2O is oxygen. Which isotope of hydrogen contains only one proton and no neutron in its nucleus. Its atomic number is 92 and its atomic mass is 238 92 14621 Electrons Protons Neutrons and Atoms.
You can take away the electron and make an ion but you cant take away any neutrons. Isotopes of hydrogen from most to least common have zero one or two neutrons. Hydrogens special structure becomes very important when you learn how hydrogen interacts.
A neutral hydrogen atom has an atomic mass of 100794 amu 1 amu. Natural hydrogen is a simple atom with one proton one electron and no neutron. Their nuclear symbols are therefore 1 H 2 H and 3 H.
Hydrogen has 1 protons and electrons in its structure. Number of neutrons. Hydrogen has no neutrons deuterium has one and tritium has two neutrons.
A normal Argon atom has 18 electrons 18 protons and 22 neutrons. How many protons electrons and neutrons does an atom with an atomic 50 and a mass 120 contain. In summary A hydrogen atom has 1 proton 1 electron 0 neutrons I hope that helps.
How many protons neutrons and electrons does a neutrally charged Hydrogen atom have. To find the amount of neutrons we take the proton number and subtract it from the mass number. That tiny little atom the tiniest of all has only one electron and one proton.
The isotopes of hydrogen have respectively mass numbers of one two and three. Most of the mass of an atom lies in its protons and neutrons. Hydrogen has three common isotopes.
It contains 8 protons and 8 electrons. The answer to the question How many neutrons does hydrogen have is zero. Protium deuterium and tritium.
Tritium contains two neutrons. That tiny little atom the tiniest of all has only one electron and one proton. Use the periodic table entry for Hydrogen to answer the question.
Argon has been found to form one neutral compound with fluorine and hydrogen called argon fluorohydride HArF. Deuterium is a hydrogen atom with an extra neutron and tritium has two extra. Only deuterium contains one neutron in its nucleus.
A normal hydrogen H atom does not have any neutrons in its tiny nucleus. Up to 24 cash back A normal hydrogen H atom does not have any neutrons in its tiny nucleus. Protium with no neutrons and deuterium with one neutron are stable isotopes while tritium with two neutrons is radioactive and undergoes decay by beta particle emission.
Hydrogen has no neutron deuterium has one and tritium has two neutrons. The isotopes of hydrogen have respectively mass numbers of one two and three. 1-10 So a neutral hydrogen atom has no neutrons.
The atoms of these isotopes have one electron to balance the charge of the one proton. Sometimes the hydrogen atom has two neutrons tritium. Hydrogen always has one proton and one neutron.
This isotope of hydrogen is the. So the atomic number is 8.

Biology The Chemistry Of Life The Chemical Foundation Of Life Atoms Isotopes Ions And Molecules The Building Blocks Oertx

Mass Of Hydrogen Atom Bartleby

How To Find The Number Of Protons Electrons Neutrons For Hydrogen H Youtube

Diagram Of A Hydrogen Atom Showing A Proton In The Nucleus And Wall Mural Murals Water Bond Structure Myloview Com
How Heavy Is One Atom Of Hydrogen Quora
Ch105 Chapter 2 Atoms Elements And The Periodic Table Chemistry
![]()
How Many Neutrons Does Hydrogen Have What S Insight

Hydrogen Atom Pictures Photos Images Of Chemistry Science For Kids Hydrogen Atom Atom Model Science For Kids
Deuterium Science Summary Presentation Page

October 14 2015 Www Mrascience Com

Hydrogen Atoms An Overview Sciencedirect Topics



Comments
Post a Comment